The Universe is conspiring to kill you. You body is under constant attack. But most days, you don't die. You don't even get sick. This is thanks to your enormously complex immune system. Full of B cells, T cells and antibodies, your immune system fights off invading armies of bacteria and viruses.
This video by Kurzgesagt explains the basics of keeping you alive. I could have used this during PY1.
Showing posts with label PY1. Show all posts
Showing posts with label PY1. Show all posts
Wednesday, August 27, 2014
Thursday, July 3, 2014
PY1 Review - Pharmaceutical Care Lab
The Pharmaceutical Care Lab (PCL) course was the only interactive class of our first year. Once a week, we would spend half the day in the lab working on a variety of topics. Instead of sitting in a stale classroom, we worked in small groups through assignments, discussions, check-offs, and compounding. I enjoyed the variety of the courses.
The most stressful part of PCL was the Top 200 Drug List. Each week we had a new list of 8-10 drugs to memorize the Generic Name, Brand Name, Therapeutic Class, and Common Indication. The weekly quiz wasn't too bad, but you did have to spell everything exactly. The Staying on Track Quiz brought about 4 weeks of drugs together and the final exam from each semester put 100 drugs into a single test. I quickly found that Quizlet offered the best method to study the drug names with rapid repetition that required correct spelling. You could study on your computer and on your phone as you rode the bus to class. For anyone that has to memorize anything, I suggest Quizlet. Below I include the Top 200 Drugs we had to learn this year. During PY2, we will use the same drugs but add much more detail including doses, side effects, and precautions.
 During each semester, we produced 4 compounded prescriptions. Sometimes a patient requires medication that is not commercially available or in a different form. Pharmacists are licensed to compound products. Some students loved compounding but others hated it. To me, it was just like any chemistry class. I liked the part. We made Benadryl solutions, Ketoprofen creams, nasal sprays, and much more. The most interesting to me was a menthol troche to soothe irritated throats. In addition to preparing the compounded drugs, we had to counsel the patient on how to use the product. I suspect more people we worried about counseling that preparation. For each product, you explained the way to take the medication, what to watch out for, and some other parts. In the profession, pharmacists can do from zero to all compounding. In the special formulations area of the hospital, compounding happens all the time. Some pharmacies do nothing but compound products for local hospitals and patients (but some got in trouble for doing too much the wrong way - there will be a post about that later). Then again, some pharmacists will never compound a products. But all pharmacists have to counsel patients on medications.
During each semester, we produced 4 compounded prescriptions. Sometimes a patient requires medication that is not commercially available or in a different form. Pharmacists are licensed to compound products. Some students loved compounding but others hated it. To me, it was just like any chemistry class. I liked the part. We made Benadryl solutions, Ketoprofen creams, nasal sprays, and much more. The most interesting to me was a menthol troche to soothe irritated throats. In addition to preparing the compounded drugs, we had to counsel the patient on how to use the product. I suspect more people we worried about counseling that preparation. For each product, you explained the way to take the medication, what to watch out for, and some other parts. In the profession, pharmacists can do from zero to all compounding. In the special formulations area of the hospital, compounding happens all the time. Some pharmacies do nothing but compound products for local hospitals and patients (but some got in trouble for doing too much the wrong way - there will be a post about that later). Then again, some pharmacists will never compound a products. But all pharmacists have to counsel patients on medications.
We began learning about Medication Therapy Management by looking a patient's med lists and searching for duplications and interactions. We took vital signs, including blood pressure. We interviewed a patient. We practiced and passed aseptic technique. The PCL covers a lot of ground. It also lets PY3s work as Teaching Assistants for the PY1s. I am looking forward to what we will do next year.
The most stressful part of PCL was the Top 200 Drug List. Each week we had a new list of 8-10 drugs to memorize the Generic Name, Brand Name, Therapeutic Class, and Common Indication. The weekly quiz wasn't too bad, but you did have to spell everything exactly. The Staying on Track Quiz brought about 4 weeks of drugs together and the final exam from each semester put 100 drugs into a single test. I quickly found that Quizlet offered the best method to study the drug names with rapid repetition that required correct spelling. You could study on your computer and on your phone as you rode the bus to class. For anyone that has to memorize anything, I suggest Quizlet. Below I include the Top 200 Drugs we had to learn this year. During PY2, we will use the same drugs but add much more detail including doses, side effects, and precautions.
 During each semester, we produced 4 compounded prescriptions. Sometimes a patient requires medication that is not commercially available or in a different form. Pharmacists are licensed to compound products. Some students loved compounding but others hated it. To me, it was just like any chemistry class. I liked the part. We made Benadryl solutions, Ketoprofen creams, nasal sprays, and much more. The most interesting to me was a menthol troche to soothe irritated throats. In addition to preparing the compounded drugs, we had to counsel the patient on how to use the product. I suspect more people we worried about counseling that preparation. For each product, you explained the way to take the medication, what to watch out for, and some other parts. In the profession, pharmacists can do from zero to all compounding. In the special formulations area of the hospital, compounding happens all the time. Some pharmacies do nothing but compound products for local hospitals and patients (but some got in trouble for doing too much the wrong way - there will be a post about that later). Then again, some pharmacists will never compound a products. But all pharmacists have to counsel patients on medications.
During each semester, we produced 4 compounded prescriptions. Sometimes a patient requires medication that is not commercially available or in a different form. Pharmacists are licensed to compound products. Some students loved compounding but others hated it. To me, it was just like any chemistry class. I liked the part. We made Benadryl solutions, Ketoprofen creams, nasal sprays, and much more. The most interesting to me was a menthol troche to soothe irritated throats. In addition to preparing the compounded drugs, we had to counsel the patient on how to use the product. I suspect more people we worried about counseling that preparation. For each product, you explained the way to take the medication, what to watch out for, and some other parts. In the profession, pharmacists can do from zero to all compounding. In the special formulations area of the hospital, compounding happens all the time. Some pharmacies do nothing but compound products for local hospitals and patients (but some got in trouble for doing too much the wrong way - there will be a post about that later). Then again, some pharmacists will never compound a products. But all pharmacists have to counsel patients on medications. We began learning about Medication Therapy Management by looking a patient's med lists and searching for duplications and interactions. We took vital signs, including blood pressure. We interviewed a patient. We practiced and passed aseptic technique. The PCL covers a lot of ground. It also lets PY3s work as Teaching Assistants for the PY1s. I am looking forward to what we will do next year.
Wednesday, July 2, 2014
Self-Care - Vitamin C
In first semester lab, each group of 3-4 students delivered a 20 minute presentation on an herbal remedy. Self-care refers to the items that customers by at the front of the store and routinely forget to inform their doctors and pharmacists about. Herbal supplements are big business making huge profits as a trendy alternative to a prescription without the risky part of actually testing the product through clinical trials. Labeled as dietary supplements, herbals do not have to actually provide data about whether or not the product works. Then again, they cannot say that the product does work either. A lot of these products rely on anecdotal evidence and the mythos of ancient, traditional or alternative medicine.
To be fair some of the products worked. A little. Most were inconclusive. And one big one - St. John's Wort - interacts with almost every pharmaceutical drug out there and can be dangerous in combinations. My group's topic was no different. We had Vitamin C.
Vitamin C, you know, occurs naturally in citrus fruits. Our body needs it and a lack of the vitamin results in Scurvy. Sailors and pirates would return from see emaciated and losing their teeth. It was determined that they lacked Vitamin C and prescribed oranges, lemons and limes. Vitamin C works great as a treatment for scurvy. But the makers of EmergenC are targeting sailors. Rather, they trick people into thinking that large amounts of Vitamin C could prevent the common cold. They even had a two-time Nobel laureate on their side. Linus Pauling loved Vitamin C taking over a 1000 mg a day or about 1667% greater than the recommended allowance. He claimed it kept him from getting sick, even when he obviously suffered from a cold. Later he claimed that it prevented cancer, even though he and his wife both died of cancer. In short the Pauling promotion of Vitamin C lingers into confusion today. Despite the assertions of the eminent scientist, no evidence ever supported him.
In our group, I landed the introductory role. My job was to get everything started and get people interested. I only presented the first five slides, but I researched and wrote one of the slides at the end (about the Cochrane Review). While I am posting this on here, I do not want to take credit on the interwebs for the entire project. Vitamin C deeply interested me and I enjoyed the history of this great confusion. My group did a great job and they should receive the credit they deserve.
Monday, June 30, 2014
PY1 Review - Pharmacotherapy
Pharmacotherapy produced divergent opinions. Some really hated the class, but other really enjoyed it. I was on the positive side and appreciated the curveballs thrown by the professor. Each semester for the remainder of the classroom portion of the PharmD program will have a therapy component. This course takes a common disorder and focuses on the way we treat it. It can't get anymore straight forward. This is the purpose of any health career, so pharmacotherapy is perhaps the most prescient class of our program (up there with PCL).
The second semester of the PY1 focuses on the #1 reason a patient encounters a health care provider - PAIN. Pain lets us know that something is wrong with our body and sends the patient to see the doctor. Everyone takes some kind of pain medicine, whether Advil and Tylenol up to Percocet and Morphine. Our task this past spring was to understand pain management.
We started by learning about medical records. Some people didn't seem to understand why. While, we do not perform the physical examination or run the clinical labs, we do have to understand what is in the file. Is the patient's potassium too high or too low? Does the positive stool guaiac have any relevance to the patient's abdominal pain? We have to be able to distinguish the pertinent positives of a medical history from pertinent negatives. This was perhaps the most confusing part to the majority of the class. I hope that my understanding can work for you. A pertinent positive refers to any abnormality in a patient's chart. If a value is outside the normal range, it is important to note. A pertinent negative refers to something that we would expect to be wrong, but is okay. So if you hypothesize that the patient has internal bleeding, but a high hematocrit that would be a pertinent negative as bleeding should decrease your blood count. Reading an understanding a chart helps the doctor diagnose a disorder, but it helps the pharmacist manage the appropriate therapy and prevent potential errors.
 Finally, we moved onto the topic of pain. We talked about metastatic pain from cancer and the various forms of joint pain from arthritis. Choosing the appropriate pain medicine depends on severity and side effects. Opioids are the best at reducing pain, but also suppress breathing. Surgical patients blow hard into a spirometer every hour to clear their lungs of potential bacteria as the morphine keeps them from doing so on their own. Patients with severe pain take a standing dose of an opioid but also need a rescue dose for breakthrough pain. The pharmacist must check the doses, convert between one opioid to the next and watch for side effects.
Finally, we moved onto the topic of pain. We talked about metastatic pain from cancer and the various forms of joint pain from arthritis. Choosing the appropriate pain medicine depends on severity and side effects. Opioids are the best at reducing pain, but also suppress breathing. Surgical patients blow hard into a spirometer every hour to clear their lungs of potential bacteria as the morphine keeps them from doing so on their own. Patients with severe pain take a standing dose of an opioid but also need a rescue dose for breakthrough pain. The pharmacist must check the doses, convert between one opioid to the next and watch for side effects.
 In the case of arthritis, pain medicines must also be used with other therapies. Also start with the lowest possible solution - Tylenol and exercise. Maybe braces could be required to stabilize the joints, but if the problem progresses, move up to NSAIDs like Advil. Unfortunately, NSAIDs cause a lot of problems like gastrointestinal bleeding, kidney problems, and clotting issues. Millions of people take NSAIDs everyday without recognizing the possible consequences. Most people take them for acute reasons like a sprain or headache, but chronic use can be a problem like with arthritis. Whether osteoarthritis, rheumatoid arthritis or gout, the disorder will affect them for the rest of their lives. Now you need treatments like allopurinol to lower the uric acid in the patient's blood to prevent gout flares. Or methotrexate or Remicade to fight the debilitation of rheumatoid arthritis. Pain can be quite complicated and the medications can cause their own problems.
In the case of arthritis, pain medicines must also be used with other therapies. Also start with the lowest possible solution - Tylenol and exercise. Maybe braces could be required to stabilize the joints, but if the problem progresses, move up to NSAIDs like Advil. Unfortunately, NSAIDs cause a lot of problems like gastrointestinal bleeding, kidney problems, and clotting issues. Millions of people take NSAIDs everyday without recognizing the possible consequences. Most people take them for acute reasons like a sprain or headache, but chronic use can be a problem like with arthritis. Whether osteoarthritis, rheumatoid arthritis or gout, the disorder will affect them for the rest of their lives. Now you need treatments like allopurinol to lower the uric acid in the patient's blood to prevent gout flares. Or methotrexate or Remicade to fight the debilitation of rheumatoid arthritis. Pain can be quite complicated and the medications can cause their own problems.
The second semester of the PY1 focuses on the #1 reason a patient encounters a health care provider - PAIN. Pain lets us know that something is wrong with our body and sends the patient to see the doctor. Everyone takes some kind of pain medicine, whether Advil and Tylenol up to Percocet and Morphine. Our task this past spring was to understand pain management.
We started by learning about medical records. Some people didn't seem to understand why. While, we do not perform the physical examination or run the clinical labs, we do have to understand what is in the file. Is the patient's potassium too high or too low? Does the positive stool guaiac have any relevance to the patient's abdominal pain? We have to be able to distinguish the pertinent positives of a medical history from pertinent negatives. This was perhaps the most confusing part to the majority of the class. I hope that my understanding can work for you. A pertinent positive refers to any abnormality in a patient's chart. If a value is outside the normal range, it is important to note. A pertinent negative refers to something that we would expect to be wrong, but is okay. So if you hypothesize that the patient has internal bleeding, but a high hematocrit that would be a pertinent negative as bleeding should decrease your blood count. Reading an understanding a chart helps the doctor diagnose a disorder, but it helps the pharmacist manage the appropriate therapy and prevent potential errors.
Thursday, June 26, 2014
PY1 Review - Pharmacodynamics
To me, this class had the wrong name. It was supposed to be an introduction to Pharmacodynamics, but became half about Pharmacokinetics. It seemed much more like an overview of general pharmacology. As an only 2nd semester course, pharmacodynamics incorporated a lot from the other courses. Overall, this was a more daunting class than our others primarily due to the tests. They were 90-minute tests crammed into 50-minute blocks. On every exam, I wrote constantly to finish just before the time ended. Luckily, the next thing was alway lunch so my hand cramps had a chance to relax.
As I said, the course wasn't really all of dynamics, but both dynamics and kinetics together. We essentially traced the path of a drug from administration through to the effect and elimination. So I will try to do a similar task. We begin after the drug has been formulated and manufactured. Let's assume a capsule of some kind. Anything given orally is called PO (per oral, by mouth). The patient takes the capsule and must pass from the GI tract into the blood stream. This isn't easy. Besides dissolution, the drug passes through the cells of the intestinal wall where it can be metabolized or it can be sent back to the GI or passed on into the blood. Once in the blood system, the portal vein takes anything absorbed by the intestines directly to the liver to be detoxified. A lot of drugs given orally to do make it past this step. Called First Pass Metabolism, any PO drug must be able to sneak through. After the liver the drug enters the systemic circulation and distributes throughout the body - hopefully to the site of action.
Drug metabolism causes a lot of problems for drug manufactures. Liver cells, hepatocytes, contain thousands of enzymes carefully designed for degrading potentially toxic substances. Most notably, the cytochrome P450 family metabolizes the vast majority of medications. CYP450 causes many problems when drugs are given in combinations. One drug could inhibit CYP450, meaning that the other drug stays in the body longer and can accumulate to toxic levels (essentially a metabolic overdose). On the other hand, the first drug could ramp up the CYP450 and decrease the amount of the other drug too fast for it to be effective. Inhibitors and Inducers must be studied carefully and considered by the pharmacist when verifying and dispensing a prescription.
If the drug avoids first pass metabolism, it must cause a desirable effect. Specifically, more positive effects than negative effects. FDA approval requires a consideration of the benefit versus the cost. Chemotherapy drugs have a lot of side effects, but the benefits outweigh the potential problems. Once
the drug reaches the site of action, the molecule binds to a cell receptor. It could turn on an action or it could turn off an action. All of these are important considerations when designing a new drug. On average, a new potential drug takes more than 10 years to reach FDA approval. 99.9% of new molecules fail to become a marketed medication.
I enjoyed the pharmacodynamics course, even if I disagreed with the title. The professors were amazing. While the exams were difficult, you had to know what you were talking about to do well. I think this course really seemed more like the introductory level of pharmacology that the other health professions take. I went to a Starbucks the other day and a lady was reading from a pharmacology textbook. During PY2, we take a pharmacokinetics course to go in much more detail. I am ready for that.
As I said, the course wasn't really all of dynamics, but both dynamics and kinetics together. We essentially traced the path of a drug from administration through to the effect and elimination. So I will try to do a similar task. We begin after the drug has been formulated and manufactured. Let's assume a capsule of some kind. Anything given orally is called PO (per oral, by mouth). The patient takes the capsule and must pass from the GI tract into the blood stream. This isn't easy. Besides dissolution, the drug passes through the cells of the intestinal wall where it can be metabolized or it can be sent back to the GI or passed on into the blood. Once in the blood system, the portal vein takes anything absorbed by the intestines directly to the liver to be detoxified. A lot of drugs given orally to do make it past this step. Called First Pass Metabolism, any PO drug must be able to sneak through. After the liver the drug enters the systemic circulation and distributes throughout the body - hopefully to the site of action.
Drug metabolism causes a lot of problems for drug manufactures. Liver cells, hepatocytes, contain thousands of enzymes carefully designed for degrading potentially toxic substances. Most notably, the cytochrome P450 family metabolizes the vast majority of medications. CYP450 causes many problems when drugs are given in combinations. One drug could inhibit CYP450, meaning that the other drug stays in the body longer and can accumulate to toxic levels (essentially a metabolic overdose). On the other hand, the first drug could ramp up the CYP450 and decrease the amount of the other drug too fast for it to be effective. Inhibitors and Inducers must be studied carefully and considered by the pharmacist when verifying and dispensing a prescription.
| FDA approval is difficult and controversial. |
the drug reaches the site of action, the molecule binds to a cell receptor. It could turn on an action or it could turn off an action. All of these are important considerations when designing a new drug. On average, a new potential drug takes more than 10 years to reach FDA approval. 99.9% of new molecules fail to become a marketed medication.
I enjoyed the pharmacodynamics course, even if I disagreed with the title. The professors were amazing. While the exams were difficult, you had to know what you were talking about to do well. I think this course really seemed more like the introductory level of pharmacology that the other health professions take. I went to a Starbucks the other day and a lady was reading from a pharmacology textbook. During PY2, we take a pharmacokinetics course to go in much more detail. I am ready for that.
Wednesday, June 25, 2014
New Drug Update - Ibruvica (ibrutinib)
During our Hospital IPPE month, we had to give a presentation on a recently approved drug from the last year. A lot of important medicines received approval including to oral treatments to Hepatitis C. I also liked that each of these drugs had been discussed during our lecture classes. I might not have remembered what they did, but I at least recognized some of the names.
I waited until the end of the sign up period, because I did not really care what I got. In the end, I selected Imbruvica, the oral treatment for mantle cell lymphoma and chronic lymphocyctic leukemia. We also had to find some way to actively engage the audience during our presentation. It's almost like I have a Master's degree in that. But really, I do. I chose to impersonate a couple of our professors from the semester. I had a pickle jar with everyone's name in it, a chemist hat, played pharmacy hangman, and wrote a short poem. I also chose a punny subtitle for the lecture. PO is the pharmacy abbreviation for ORAL - tablet, capsule, etc.
I waited until the end of the sign up period, because I did not really care what I got. In the end, I selected Imbruvica, the oral treatment for mantle cell lymphoma and chronic lymphocyctic leukemia. We also had to find some way to actively engage the audience during our presentation. It's almost like I have a Master's degree in that. But really, I do. I chose to impersonate a couple of our professors from the semester. I had a pickle jar with everyone's name in it, a chemist hat, played pharmacy hangman, and wrote a short poem. I also chose a punny subtitle for the lecture. PO is the pharmacy abbreviation for ORAL - tablet, capsule, etc.
Monday, June 23, 2014
PY1 Review - Pharmaceutics
As the keepers of tablets, capsules and suppositories, pharmacists must obviously understand how these products are manufactured and deliver medicines to the body. That class is called pharmaceutics and is unique to the field of pharmacy. We took two semesters worth of information for one of the most interesting classes we had during the PY1 year.
Determining the route of administration of a drug is just as important as choosing the right drug. If a person has diabetes, as many people do, we want to give them insulin. A lot of other medicines work as well, but insulin remains the standard. So how do you get the insulin into the body? Most people know about insulin injections - a subcutaneous administration of an endogenous protein. But 3-4 injections a day adds up quickly to 21-28 a week or 1,095-1,460 injections per year. What if we could devise a better way to deliver insulin without a needle? That is pharmaceutics.
The most common method of taking medicines today are pills. Pills, pills, pills. Patients taking 30 or 40 per day. Tablets can be different sizes and different shapes. The type of oral medication depends on the aspects of the drug itself. Some drugs degrade in low pH, like in the stomach. In that case, you don't want the drug released until it gets into the small intestines. Enteric coating can protect drug by not dissolving in the stomach. A lot of science goes into designing the right pill. A lot more goes into making the pill stable as some sit in boxes for 2 years from manufacture to your mouth. We talked a lot about the excipients added to medications and the testing process each product must go through.
Unfortunately, great pills don't always deliver all of the medicine through the digestive system. The amount that actually gets through to your body determines the bioavailability. Some oral products have very low bioavailability, but the gold standard will always be intravenous injection. If pills are complicated to make, IVs have their own problems. Producing IV meds involves a lot of thought in stability and sterility. Hospitals have giant clean rooms to prepare IVs to reduce the likelihood of contamination. Even then, a drug in saline solution may only be stable for a few hours before precipitating into a useless cloudy mess.
I really liked both semesters of pharmaceutics. The topics were interesting and the wide range of professors provided a lot of entertainment. Pharmaceutics seems like one of the secrets kept by the pharmacists from the other health care professionals. You can take this subject with you to counseling patients in the community, to preparing IV bags in a hospital, or to developing a new billion dollar drug in the industry.
Determining the route of administration of a drug is just as important as choosing the right drug. If a person has diabetes, as many people do, we want to give them insulin. A lot of other medicines work as well, but insulin remains the standard. So how do you get the insulin into the body? Most people know about insulin injections - a subcutaneous administration of an endogenous protein. But 3-4 injections a day adds up quickly to 21-28 a week or 1,095-1,460 injections per year. What if we could devise a better way to deliver insulin without a needle? That is pharmaceutics.
The most common method of taking medicines today are pills. Pills, pills, pills. Patients taking 30 or 40 per day. Tablets can be different sizes and different shapes. The type of oral medication depends on the aspects of the drug itself. Some drugs degrade in low pH, like in the stomach. In that case, you don't want the drug released until it gets into the small intestines. Enteric coating can protect drug by not dissolving in the stomach. A lot of science goes into designing the right pill. A lot more goes into making the pill stable as some sit in boxes for 2 years from manufacture to your mouth. We talked a lot about the excipients added to medications and the testing process each product must go through.
Unfortunately, great pills don't always deliver all of the medicine through the digestive system. The amount that actually gets through to your body determines the bioavailability. Some oral products have very low bioavailability, but the gold standard will always be intravenous injection. If pills are complicated to make, IVs have their own problems. Producing IV meds involves a lot of thought in stability and sterility. Hospitals have giant clean rooms to prepare IVs to reduce the likelihood of contamination. Even then, a drug in saline solution may only be stable for a few hours before precipitating into a useless cloudy mess.
I really liked both semesters of pharmaceutics. The topics were interesting and the wide range of professors provided a lot of entertainment. Pharmaceutics seems like one of the secrets kept by the pharmacists from the other health care professionals. You can take this subject with you to counseling patients in the community, to preparing IV bags in a hospital, or to developing a new billion dollar drug in the industry.
Thursday, June 19, 2014
Irinotecan - Topoisomerase Poison
Easily the most stress of PY1 came from the Biochemistry Presentations. Another group lab presentation, but instead of fun, herbal supplements, we talked about the way chemo and antiviral medications worked. I greatly enjoyed the topic, but the level of difficulty increased dramatically. My group was given irinotecan, a treatment for colorectal cancer. Not a lot of jokes were going to be made in this one.
Irinotecan is a very interesting drug. The molecule blocks the effects of Topoisomerase 1 and results in an accumulation of double strand breaks that leads to cell death. Of the cancers, Topo1 increases the most in colorectal cancer, but it is being investigated for others as well. Irinotecan acts as a prodrug for its active metabolite, SN-38, which undergoes glucuronidation and can become a part of enterohepatic recycling leading to extreme diarrhea. The complexity of irinotecan showed up in every class during the spring semester. Which was great, as I became an expert on the drug.
While other groups took the traditional lecture approach to the presentation, my group wanted to do something different. We created a demonstration of Topoisomerase with rope, scissors and duct tape. As DNA is copied for cell division, the unwinding of the helix increases the stress on the molecule upstream. Topo1 releases that stress by cutting one strand of the DNA, allowing the molecule to unravel, and resealing the original cut. Pretty simple idea, especially if you could have seen our demonstration. We did do it, but due to snow delays only six people were in the room and I doubt anyone watched the video afterward. However, it was a big hit with the professors and TAs as other people who weren't there came up and talked to me about it.
As I said, it was a group project with a great group of girls. I do not want to take all the credit by posting it on her. My job was the Mechanism of Action. I chose it specifically because it generally received the most difficult questions from the feared professor. Other than issues with the weather, our group nailed the presentation - possibly even the best of the semester.
Irinotecan is a very interesting drug. The molecule blocks the effects of Topoisomerase 1 and results in an accumulation of double strand breaks that leads to cell death. Of the cancers, Topo1 increases the most in colorectal cancer, but it is being investigated for others as well. Irinotecan acts as a prodrug for its active metabolite, SN-38, which undergoes glucuronidation and can become a part of enterohepatic recycling leading to extreme diarrhea. The complexity of irinotecan showed up in every class during the spring semester. Which was great, as I became an expert on the drug.
While other groups took the traditional lecture approach to the presentation, my group wanted to do something different. We created a demonstration of Topoisomerase with rope, scissors and duct tape. As DNA is copied for cell division, the unwinding of the helix increases the stress on the molecule upstream. Topo1 releases that stress by cutting one strand of the DNA, allowing the molecule to unravel, and resealing the original cut. Pretty simple idea, especially if you could have seen our demonstration. We did do it, but due to snow delays only six people were in the room and I doubt anyone watched the video afterward. However, it was a big hit with the professors and TAs as other people who weren't there came up and talked to me about it.
As I said, it was a group project with a great group of girls. I do not want to take all the credit by posting it on her. My job was the Mechanism of Action. I chose it specifically because it generally received the most difficult questions from the feared professor. Other than issues with the weather, our group nailed the presentation - possibly even the best of the semester.
Wednesday, June 18, 2014
PY1 Review - Biochemistry
Out of all the classes on our PY1 schedule, biochemistry frightened me the most. While I have taken biology and I have taken chemistry, I had never been enrolled in a class that combined the two. Other PharmD students had majored in biochemistry, and I had absolutely no idea what to think. By the second class, my fears were realized as I sat through a 90 minute presentation without understanding a single word. Biochem would be my downfall in pharmacy school.
Actually, biochemistry turned out to be one of my favorite classes. Many of the topics had been covered in the high school bio courses I taught or at the very least extended from the same concepts. If I had really known about biochemistry during my undergraduate years, I may have ended up majoring in it and began pharmacy school 10 years sooner! In pharmacy school, biochemistry serves as the prerequisite to the medicinal chemistry on PY2. Two semesters divide the topic into nucleic acids and not-nucleic acids.
The first semester began as a review of organic chemistry followed by the basics of proteins,
carbohydrates, and lipids. From organic, you should know all of the functional groups names and relative pKas. Under proteins, the course covers some about structure, but mostly enzymatic kinetics. Michaelis-Menten mechanics drive every theory about enzymes and drugs. For carbohydrates, you must memorize the catabolic processes of glycolysis, Krebs, and electron transport chain and the anabolic processes including gluconeogenesis. Finally, lipids cover the basic structures of triglycerides and phospholipids. If it sounds like a lot of information, it is. Those that majored in biochem sailed easily through this part. Others, like me, had to do a lot of work to keep up and it didn't get any easier next semester.
In the second part of biochemistry, we studied anything and everything related to nucleic acids. I was really interested in this part as many antibiotics target the DNA differences between us and them.
Oncology also attacks the properties of our own DNA to kill rapidly growing cells. This course began with the basics of DNA, similar to what I taught to my classes but again with a bit more detail.
Once through the discoveries of Watson and Crick, we went over the way cells use the DNA template for replication, transcription and translation. Really, the order of topics should be familiar to anyone. The difficulty came from the depth of information covered and the density of exam questions. Compared to the first semester, we talked about all kinds of drugs affecting DNA, RNA, and ribosomes. I know more about Topoisomerase 1 than I think I will ever use, but I understand the importance of irinotecan in the treatment of colorectal cancer. Success in Biochem 2 required a lot of work, but it was worth it in the end.
I feel good about my understanding of biochemistry after this year. I won't be earning a PhD in the topic, but I can hold my own. Will it help my career as a pharmacist? I think so. That really determines which direction you go. While you may not use gluconeogenesis, you do have to know that cortisol (and any other prescription steroid) stimulates the process. Otherwise it varies. Oncologists, yes. Community, maybe not. Overall, it broadens your knowledge base and provides more opportunities for you to pursue after pharmacy school ends.
Actually, biochemistry turned out to be one of my favorite classes. Many of the topics had been covered in the high school bio courses I taught or at the very least extended from the same concepts. If I had really known about biochemistry during my undergraduate years, I may have ended up majoring in it and began pharmacy school 10 years sooner! In pharmacy school, biochemistry serves as the prerequisite to the medicinal chemistry on PY2. Two semesters divide the topic into nucleic acids and not-nucleic acids.
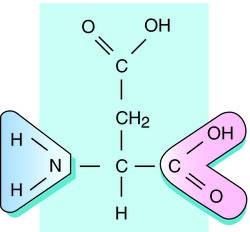 |
| Aspartate - structure with acidic side chain |
carbohydrates, and lipids. From organic, you should know all of the functional groups names and relative pKas. Under proteins, the course covers some about structure, but mostly enzymatic kinetics. Michaelis-Menten mechanics drive every theory about enzymes and drugs. For carbohydrates, you must memorize the catabolic processes of glycolysis, Krebs, and electron transport chain and the anabolic processes including gluconeogenesis. Finally, lipids cover the basic structures of triglycerides and phospholipids. If it sounds like a lot of information, it is. Those that majored in biochem sailed easily through this part. Others, like me, had to do a lot of work to keep up and it didn't get any easier next semester.
 |
| Irinotecan - mechanism of action. |
Oncology also attacks the properties of our own DNA to kill rapidly growing cells. This course began with the basics of DNA, similar to what I taught to my classes but again with a bit more detail.
Once through the discoveries of Watson and Crick, we went over the way cells use the DNA template for replication, transcription and translation. Really, the order of topics should be familiar to anyone. The difficulty came from the depth of information covered and the density of exam questions. Compared to the first semester, we talked about all kinds of drugs affecting DNA, RNA, and ribosomes. I know more about Topoisomerase 1 than I think I will ever use, but I understand the importance of irinotecan in the treatment of colorectal cancer. Success in Biochem 2 required a lot of work, but it was worth it in the end.
I feel good about my understanding of biochemistry after this year. I won't be earning a PhD in the topic, but I can hold my own. Will it help my career as a pharmacist? I think so. That really determines which direction you go. While you may not use gluconeogenesis, you do have to know that cortisol (and any other prescription steroid) stimulates the process. Otherwise it varies. Oncologists, yes. Community, maybe not. Overall, it broadens your knowledge base and provides more opportunities for you to pursue after pharmacy school ends.
Thursday, June 12, 2014
Tigecycline - A New Weapon Against Antibacterial Resistance
As soon
as Alexander Fleming accidentally discovered the medicinal benefit of
antibiotics, bacteria began fighting back.
Penicillin appeared as the kind of “magic bullet” Paul Ehrlich
envisioned, but the ability of microbes to evolve resistance mechanisms has led
to an evolutionary and pharmacological arms race over the last century. Antibiotics attack susceptible bacteria
through specific mechanisms based on the cellular and metabolic differences
between prokaryotes and eukaryotes. One
class of antibiotics, the tetracyclines, prevents the synthesis of new proteins
by binding to the bacterial ribosomes.
Unfortunately, the bacteria responded by modifying their ribosomes and
pumping the antibiotic out of the cell.1 Tigecycline, a glycycline built of the
tetracycline scaffold, possesses a unique functional group that allows it to
block the bacterial ribosome and simultaneously evade bacterial resistance
mechanisms.2 Developed under
the name GAR-936 by Wyeth Pharmaceuticals in Philadelphia, tigecycline (brand
name: Tygacil) provides a new weapon in the rapidly changing battle of
antibacterial resistance.
Tetracyclines
and tigecycline specifically target the 30S subunit of the bacterial ribosome
by binding reversibly to the A-site, and preventing the accommodation of the
tRNA.3 Within the 30S
subunit, tetracylines interfere with the H34 helical region of the 16S rRNA
through hydrogen bonds between the hydrophilic parts of the drug and the
phosphate backbone. The resulting steric
hindrance prevents aminoacyl-tRNA from pivoting to the P-site and blocks the
elongation of the peptide chain. While
tetracyclines bind to the subunit in one direction, tigecycline blocks the
A-site although through a different orientation. This unique binding increases
tigecycline’s affinity almost 5-fold giving the antibiotic its bacteriostatic
properties and a method of circumventing the resistance proteins in some
strains of bacteria.2
All
antibiotic products face the challenge of bacterial resistance. Formerly susceptible bacterial strains employ
two major resistance mechanisms against tetracycline antibiotics. First, bacteria up-regulate efflux pumps to
expel the harmful antibiotics from the cell before it can perform its
inhibitory activity. When Gram-negative
bacteria encounter a tetracycline product, efflux genes known in sequence as TetA-E turn on to a decrease the
concentration of drug within the cell.
Gram-positive bacteria remove tetracyclines through the same efflux
process, but with the genes TetG-L. These gram-positive bacteria also produce
extra small proteins that block the location of tetracycline binding through
the genes TetM or TetO.1,4 The protective proteins compete with the
tetracycline and reduce the effectiveness of the drug. Researchers designed tigecycline with the
intention of avoiding the Tet efflux
pumps by including a bulky side chain that would stay in the cell to continue
working, but they also found that it continued to work in the presence TetM proteins
During
development in the early 1990’s, tigecycline became the main focus of a new
antibiotic class of glycyclines that retained the ribosome blocking mechanisms
of tetracycline but avoided the evolved Tet
resistance.4 Starting with
tetracycline’s four-ring structure, researchers developed new generations of
antibiotics by attaching various functional groups to evade efflux and avoid TetM
proteins. For example, doxycycline moves
a hydroxyl group from the C-ring to the B-Ring, while minocycline includes a
dimethylamine to the 7-position on the D-ring.
Minocycline proved to be a strong antibiotic,
but eventually succumbed to similar mechanisms of resistance. Researchers attempted to improve minocycline
by including a new functional group to C-9 position of the D-ring. An early glycycline,
dimethylglycylamido-minocycline (DMG-MINO) provided a hopeful avenue, but tigecycline
showed the most promise against the more clinically relevant, multidrug
resistant pathogens.5,6 Structurally, tigecycline, a chemical
descendant of minocycline, adds a tert-butyl-glycylamido group to the D-Ring,
which provides the anti-resistance benefits while maintaining antibacterial
properties with three main improvements.
First, the molecule became more lipophilic allowing for easier transport
into the cell. Because tetracyclines act
on ribosomes, they must reach the cytosol to perform their inhibitory
mechanisms. The additional side chain
increases tigecycline’s volume of distribution to 7-9 L/kg, showing that more
of the drug resides tissues than in plasma.3 Secondly, the extra-long alkyl group creates
enough steric hindrance to prevent efflux from the cell. Where tetracyclines would be removed, the
bulky tigecycline continues to work.
Thirdly, tigecycline shows five times more affinity to the 30S ribosome compared
to tetracycline.2 The
9-tert-butyl-glycylamido group shifts the orientation of tigecycline within the
30S ribosome and generates more stable hydrogen bond stacking, shielding the
molecule from TetM protective
proteins.7,8 The important glycylamido
addition to the minocycline backbone provides the biochemical opportunity for
the pharmacological improvements of tigecycline over minocycline and other
tetracyclines. Unfortunately, some
anaerobic bacteria have displayed resistance to tigecycline through the upregulation
of a different set of efflux pumps. Enterobacter and Acinetobacter increase the number of RND pumps on their membranes,
which are not blocked by the bulky side chain and remove tigecycline from the
cell.2
Interest
in tigecycline led to worldwide testing process as many countries sponsored
research into the effectiveness of tigecycline through the TEST Program
(Tigecycline Evaluation and Surveillance Trial). Tigecycline showed similar or improved
results to treatment by minocycline (the closest relative), to a vancomycin-azetrenam
combination, and to an imipenem-cilastatin combination. Specifically, tigecycline provides
hope to treat the various drug-resistant strains like vancomycin-resistant Enterococci (VRE), methicillin-resistant
Staphalococcus aureus (MRSA),
penicillin-resistant Streptococcus
pneumonia, and beta-lactamase producing E.
coli. However, the TEST program has
shown regional variations in the activities of tigecycline against resistant
bacteria and a growing resistance to tigecycline itself. The FDA approved tigecycline for the
treatment of complicated intra-abdominal infections, community-acquired
bacterial pneumonia, and complicated skin infections against a wide range of
species.3,4,9 The FDA Black Box Warning suggests a 0.6% increase in
morality risk and advises the use of tigecycline only when alternatives are not
available.9
Unsusceptible
to the normal methods of tetracycline resistance, tigecycline continues to
inhibit the 30S ribosomal subunit despite Tet efflux transporters or TetM proteins that protect the
accommodation site of the ribosome. Tigecycline demonstrates the creative
ability biochemistry provides to overcome bacterial resistance by building on
the structure of minocycline. Tigecycline’s bulky side chain generates the
improved characteristics of tigecycline.
At this point, tigecycline remains the only FDA approved glycycline and
offers a potent weapon against antibacterial resistance.
References
1. Projan S.
Preclinical pharmacology of GAR-936, a novel glycycline antibacterial agent. Pharmacotherapy.
2000;20(9):219-223.
2. Seputiene V,
Povilonis J, Armalyte J, Suziedelis K, Pavilonis A, Suziedeliene E. Tigecycline
- how powerful is it in the fight against antibiotic-resistant bacteria? Medicina
(Kaunas). 2010;46(4):240-248.
3.
Doan T, Fung H,
Mehta D, Riska P. Tigecycline: A glycylcyline antimicrobial agent. Clin
Thera. 2006;28(8):1079-1106.
4.
Peterson L. A
review of tigecycline - the first glycycline. International Journal of
Antimicrobial Agents. 2008;32(S4):S215-S222.
5.
Garrison M, Neumiller
J, Setter S. Tigecycline: An investigational glycycline antimicrobial with
activity against resistant gram-positive organisms. Clin Thera.
2005;27(1):12-22.
6.
Loh E,
Ellis-Grosse E, Petersen P, Sum P, Projan S. Tigecycline: A case study. Expert
Opin Drug Discov. 2007;2(3):403-418.
7.
Olson M, Ruzin
A, Feyfant E, Rush T, O'Connell J, Bradford P. Functional, biophysical, and
structural bases for antibacterial activity of tigecycline. Antimicrob
Agents Chemother. 2006;50(6):2156-2166.
8.
Jenner L, Starosta
A, Terry D, et al. Sturctural basis for potent inhibitory activity of the
antibiotic tigecycline during protein synthesis. PNAS.
2013;110(10):3812-3816.
9.
Tygacil [package
insert]. Philadelphia, PA: Wyeth Pharmaceuticals Inc; September 2013.
Monday, June 9, 2014
PY1 Review - Physiology
Starting pharmacy school marked a major shift in my life. No longer do I stand in front of the class talking about science and lighting things on fire. Now I sit in the front row, taking notes and answering questions. Before a couple years ago, pharmacy school was never an option that crossed my mind. Not because I did not think that I could do it, but because I had never learned anything about it. Teachers, counselors, nor professors ever presented pharmacy as a potential option. I hope to change some of that. Obviously, I am attempting to write on this website and twitter, but I also hope to talk to teachers and students about an exciting career path.
In attempt to help, I thought that I would review some of my experiences in the first year of pharmacy school. My first topic will be Physiology.
 Since I taught anatomy, I figured physiology would be one of my advantages this year. But the way I taught the course was completely different from my first course. In high school anatomy, most teachers begin with an overview of terms then on to cells, tissues, skin, bones, muscles, brain, endocrine, and whatever else can fit in before the final exam. The pharmacy physiology class began with Cardiology. It makes sense. Heart attacks and congestive heart failure are among the biggest killers in the United States. Next to pain, medicines to control blood pressure top the most prescribed and sold drugs each year. HCTZ and other diuretics balance the accumulation of water while ACE inhibitors and statins affect the diameter of blood vessels. Rather than primarily focusing on the structural anatomy, pharmacy physiology appropriately addresses the function of the body.
Since I taught anatomy, I figured physiology would be one of my advantages this year. But the way I taught the course was completely different from my first course. In high school anatomy, most teachers begin with an overview of terms then on to cells, tissues, skin, bones, muscles, brain, endocrine, and whatever else can fit in before the final exam. The pharmacy physiology class began with Cardiology. It makes sense. Heart attacks and congestive heart failure are among the biggest killers in the United States. Next to pain, medicines to control blood pressure top the most prescribed and sold drugs each year. HCTZ and other diuretics balance the accumulation of water while ACE inhibitors and statins affect the diameter of blood vessels. Rather than primarily focusing on the structural anatomy, pharmacy physiology appropriately addresses the function of the body.
This particular course was set up differently from the others. Before the unit, we read a document that reviewed the system and then took a quiz. We answered the quiz questions individually and then as a group. In the next class, the ever changing professors (clinicians in the specific field) taught through case studies. Before exams, we faced cases that integrated several systems together. Despite the strange course set up, I felt that I learned quite a lot. My knowledge base as an anatomy teacher definitely helped, but would not have carried me through the course. I really like learning anatomy through the lens of medications that alter or restore baselines.
For any teachers interested, I will list the order topics here.
Module 1 - Cardiology
Module 2 - Renal
Module 3 - Hematology
Module 4 - Gastrointestinal
Module 5 - Central Nervous System
Module 6 - Respiratory
Module 7 - Endocrine
Obviously, not much discussion about skin, bones and muscles. Instead, the course featured the primary health care related systems. Millions of people live on blood pressure medications and kidney problems drastically affect a patient's response to drugs. How many people suffer from acid reflux or take mood stabilizing medications. I like the idea of teaching the human body through clinical importance instead of easy memorization and textbook order. If I had really thought about it when I was teaching, I would have strongly supported this curriculum. Like 25% of the students take some sort of ADHD medication - Concerta, Adderall, Strattera. Many kids suffer from asthma taking Proventil, Combivent, Advair. Maybe more high school anatomy teachers can bring a clinical pharmacy perspective to the classroom.
In attempt to help, I thought that I would review some of my experiences in the first year of pharmacy school. My first topic will be Physiology.
 Since I taught anatomy, I figured physiology would be one of my advantages this year. But the way I taught the course was completely different from my first course. In high school anatomy, most teachers begin with an overview of terms then on to cells, tissues, skin, bones, muscles, brain, endocrine, and whatever else can fit in before the final exam. The pharmacy physiology class began with Cardiology. It makes sense. Heart attacks and congestive heart failure are among the biggest killers in the United States. Next to pain, medicines to control blood pressure top the most prescribed and sold drugs each year. HCTZ and other diuretics balance the accumulation of water while ACE inhibitors and statins affect the diameter of blood vessels. Rather than primarily focusing on the structural anatomy, pharmacy physiology appropriately addresses the function of the body.
Since I taught anatomy, I figured physiology would be one of my advantages this year. But the way I taught the course was completely different from my first course. In high school anatomy, most teachers begin with an overview of terms then on to cells, tissues, skin, bones, muscles, brain, endocrine, and whatever else can fit in before the final exam. The pharmacy physiology class began with Cardiology. It makes sense. Heart attacks and congestive heart failure are among the biggest killers in the United States. Next to pain, medicines to control blood pressure top the most prescribed and sold drugs each year. HCTZ and other diuretics balance the accumulation of water while ACE inhibitors and statins affect the diameter of blood vessels. Rather than primarily focusing on the structural anatomy, pharmacy physiology appropriately addresses the function of the body.This particular course was set up differently from the others. Before the unit, we read a document that reviewed the system and then took a quiz. We answered the quiz questions individually and then as a group. In the next class, the ever changing professors (clinicians in the specific field) taught through case studies. Before exams, we faced cases that integrated several systems together. Despite the strange course set up, I felt that I learned quite a lot. My knowledge base as an anatomy teacher definitely helped, but would not have carried me through the course. I really like learning anatomy through the lens of medications that alter or restore baselines.
For any teachers interested, I will list the order topics here.
Module 1 - Cardiology
Module 2 - Renal
Module 3 - Hematology
Module 4 - Gastrointestinal
Module 5 - Central Nervous System
Module 6 - Respiratory
Module 7 - Endocrine
Obviously, not much discussion about skin, bones and muscles. Instead, the course featured the primary health care related systems. Millions of people live on blood pressure medications and kidney problems drastically affect a patient's response to drugs. How many people suffer from acid reflux or take mood stabilizing medications. I like the idea of teaching the human body through clinical importance instead of easy memorization and textbook order. If I had really thought about it when I was teaching, I would have strongly supported this curriculum. Like 25% of the students take some sort of ADHD medication - Concerta, Adderall, Strattera. Many kids suffer from asthma taking Proventil, Combivent, Advair. Maybe more high school anatomy teachers can bring a clinical pharmacy perspective to the classroom.
Wednesday, June 4, 2014
Intranasal-to-Brain Drug Delivery - A Review
Neurodegenerative diseases
impact the lives of patients and their families. As the brain slowly deteriorates, patients
lose control of their bodies, thoughts, and ultimately succumb to the disease. An aging population generates a higher incidence
of Alzheimer’s disease, Parkinson’s disease, and other neurological
disorders. Concurrently, the rise of
Autism Spectrum disorders has become an interesting area of potential
research. As of now, neurological medicines
only treat the symptoms and rarely address the root cause – dying or misfiring neurons. Getting drugs into the brain has proven a difficult
task because of the impermeable blood-brain barrier (BBB). Newly developed tools circumvent the BBB by
going through the nose. Intranasal-to-brain
(INB) delivery provides a hopeful avenue to slow, treat, and prevent the progression
of the diseases of the central nervous system (CNS) with greater efficacy, less
invasion and reduced toxicity. Medical
professionals should recognize the difficulty of delivering drug to the brain
and the need for easier, direct route such as nasal administration.
The Blood Brain
Barrier
As the controlling organ of
the body, the brain is locked behind two major defense mechanisms. The cranium provides a hard case that limits
access to only highly invasive techniques that inject drugs through the skull
directly into the parenchyma or cerebrospinal fluid. The BBB internally protects the CNS from potentially
damaging foreign chemicals travelling through the bloodstream. Toxins and potential medications are blocked
from neurons by several mechanisms. Despite
the rich network of blood vessels reaching all areas of the brain, the endothelial
lining of the capillaries are fused together with extremely tight junctions
preventing the passage of many molecules.1 Only the smallest hydrophilic molecules can
enter the brain through the paracellular route around the tight junctions. Some small, lipophilic molecules can cross
through the epithelial cells via the transcellular route.2 Prodrugs can increase lipophilicity and
nanoparticles can hide polar regions inside a PLGA liposome. Larger endogenous molecules are selectively
allowed across the barrier through transport proteins, like insulin and
transferrin, which we can use to transport drugs across.1 While we can make drugs lipophilic or hijack
transporter proteins to cross the membrane, P-gp efflux pumps work to actively
remove foreign compounds. Some drugs can
be given with P-gp inhibitors to increase the chances of remaining inside the
brain.3 All of these defense
mechanisms make getting drugs into the brain incredibly difficult. Indeed, as little as 1% of an intravenous
dose may reach and remain within the CNS.
The larger doses required leads to toxic side effects elsewhere in the
body.4 To treat the growing
problem of neurodegenerative disorder, we need a less invasive, patient
directed route of administration that provides rapid onset of action, higher
brain concentrations, and lower systemic distribution and side effects. Going through the nose to the olfactory bulb
provides each of our requirements.
The Nasal Anatomy
The nasal cavity performs
three functions for the human body: warms air, filters large particles, samples
odors. In direct contact with the
external environment, the cavity is lined with mucosal epithelium. Mucus, secreted from surrounding glands,
traps foreign bodies, while the cilia continuously push the mucus to the esophagus
for elimination. Any nasal medication
must be absorbed quickly or be washed into the stomach. When we breathe, air enters through the nares,
past the vestibule and into a large nasal cavity. The empty space contains specialized folds,
called conchae, which increase the surface area and create turbulence to help
warm the air on the way to the lungs. Most
nasal sprays for the treatment of rhinitis target the inferior and middle
conchae. The rich vasculature in the area provides access to systemic
circulation, but effects are limited due to nasal mucus. To target the CNS however, specialized INB
sprays target the superior concha to directly access the olfactory bulb. Comprised of millions of nerve endings, the
olfactory bulb sends out those nerve endings through the holes in the cribriform
plate to test the air for smells and provides a pathway from the nasal cavity
to the brain. As such, the nasal cavity
is a rare location with open access to the nervous system. INB drugs can bypass the difficult BBB treat
CNS disorders.5
Reaching the Brain
through the Nose
To reach our goal, INB drugs
do not target the vasculature, but rather the top of the nasal cavity –
olfactory bulb and cribriform. If
absorbed systematically, the drug would still have to pass through the BBB
reducing the neuronal availability and potentially cause toxicity. INB drugs avoid systemic circulation by
entering the brain through the more permeable epithelial membrane above the
superior concha. Any INB drug reaches
the CNS through two main pathways. The
epithelial pathway uses paracellular transport around the olfactory epithelium,
across the cribriform plate and into the subarchnoid space. The CSF carries the drug particle throughout
the brain or clears into the systemic circulation.6 Using the epithelial pathway requires a
small, hydrophilic molecule. An
olfactory pathway uses the nerve endings to internalize the drug and ferry it
to the olfactory bulb and into the olfactory region of the brain. These molecules must trigger endocytosis to
promote transport.
While easier to reach than
through the blood, the nasal cavity still possesses a number of factors that
can affect absorption. As stated before,
small molecular weight drugs are ideal as absorption is limited at 1000 daltons. The secretion and removal of mucus can affect
absorption clearing drug from the site of absorption. Few enzymes exist in the area, but
degradation remains a concern for drug stability, especially for proteins. Finally, rhinitis can alter absorption.5 Still, the nasal cavity remains a valid
alternative to administer drugs to the CNS, especially if the drug is orally
ineffective, blocked by the BBB, or requires a rapid onset of action. Several modifications can protect from these
protections and improve INB delivery.
To improve uptake through the
olfactory bulb, either the drug or the formulation can be modified. Prodrugs take advantage of local enzymes and
increase lipophilicity. Excipients
transiently open up mucosal pores to increase absorptions. Chitosan is a natural polysaccharide that
binds to the mucosal membrane and loosens the tight junctions between
epithelial cells allowing more drug to enter.5 PLGA nanoparticles can increase absorption by
attaching a lectin ligand, triggering receptor-mediated endocytosis.7 Currently, several drugs are being designed
and studied to use these methods to treat a variety of CNS disorders. Mouse models show an increase in brain
distribution through INB relative to normal intravenous administration, opening
the door to potential treatments for Alzheimer’s disease and autism.
Potential
Treatments through INB
Alzheimer’s patients face a
dark diagnosis with little positive light.
Chi Zhang and his group in Shanghi hope to make the outlook a little
brighter. Basic fibroblast growth factor
(bFGF) simulates the growth of neurons, but remains blocked by the BBB. Intravascular delivery of the peptide shows
that only 1% reaches the brain. INB offers
an alternative route but must overcome mucusal clearance and protein
degradation. Incorporating bFGF into PEG-PLGA
nanoparticles coated with Solanum
tuberosum lectin, Zhang’s group showed increased residence time, CNS
concentration of bFGF in Alzheimer-model rats and corresponding improvement in
memory tests. Direct bFGF administration
to the brain through the nose decreases plasma concentrations resulting in
fewer toxicities.7
Each day, more children
receive diagnoses of autism spectrum disorders, but few treatments exist. Evdokia Anagnostou and her Toronto-based lab
want to administer intranasal oxytocin as a potential cure. Children with ASD have poor social skills,
tend to perform repetitive behaviors, and have reduced levels of oxytocin in the
blood. Medical oxytocin could hopefully ameliorate
many autism symptoms. Because oxytocin degrades
in the intestines by chymotrypsin, it cannot be given orally. Intravascular injection of oxytocin shows
positive effects, but is too invasive for children. INB produced better CNS distribution with
easier administration. While early in
the process, children treated with INB oxytocin showed promising improvements
in autism-related behaviors.8
Conclusion
Getting medication into the
brain remains a difficult task as many molecules fail to reach the desired
destination. The BBB protects the
neurons but makes treatment more difficult.
As the population grows and ages, more patients will be diagnosed with
neurodegenerative diseases, like Alzheimer’s.
A targeted nasal spray to the olfactory bulb could potentially deliver
therapeutic compounds for a variety of CNS disorders. INB is a rapid acting route that avoids the
restrictive BBB and potentially toxic systemic side effects. More studies must determine the safety of
this route on a larger scale and show more examples of efficacy. We should learn more about local side effects
of penetration enhancers on an important protective membrane or sensitivity in
the nasal cavity leading to rhinitis. Overall,
INB offers a safer, less invasive route of administration directly to the brain
and can potentially help the patients with the frightening diseases of the CNS.
References
1.
Patel M, Goyal B, Bhadada S, Bhatt J, Amin A. Getting into the brain:
Approaches to enhance brain drug delivery. CNS Drugs. 2009;1:35-58.
2.
Illum L. Nasal drug delivery: New developments and strategies. DDT.
2002;7(23):1184-1189.
3.
Merkus F, van den Berg P. Can nasal drug delivery bypass the
blood-brain barrier? questioning the direct transport theory. Drugs R D.
2007;8(3):133-144.
4.
Goldsmith M, Abramovitz L, Peer D. Precision nanomedicine in
neurodegenerative disease. Asc Nano. 2014;8(3):1958-1965.
5.
Tayebati S, Nwankwo I, Amenta F. Intranasal drug delivery to the
central nervous system: Present status and future outlook. Curr Pharm Design.
2013;19:510-526.
6.
Illum L. Nasal drug delivery - possibilities, problems and solutions. J
Controlled Release. 2003;87(187):198.
7.
Zhang C, Chen J, Feng C, et al. Intranasal nanoparticles of basic
fibroblast growth factor for brain delivery to treat alzheimer's disease. Int
J Pham. 2014;461:192-202.
8.
Anagnostou E, et al. Intranasal oxytocin in the treatment of autism
spectrum disorders: A review of literature and early safety and efficacy data
in youth. Brain Res. 2014.
Subscribe to:
Comments (Atom)